Day 11: Maillard Reaction Fundamentals – Notes
Topic 1: How Sugars + Amino Acids Create Flavor
Page 1/3: The Chemistry of Non-Enzymatic Browning
- Maillard Reaction Definition: A complex series of non-enzymatic chemical reactions between reducing sugars (e.g., glucose, fructose) and amino acids (the building blocks of proteins). It is the primary reaction responsible for the brown color, savory depth, and much of the aroma in roasted coffee, baked bread, seared meat, and toasted grains.
- Initial Reactants in Coffee:
- Sugars: Sucrose is the most abundant sugar in green coffee. During the early stages of roasting (drying), some sucrose is broken down into its reducing sugar components: glucose and fructose. These are the active participants.
- Amino Acids: Green coffee contains approx4% protein and free amino acids (e.g., Proline, Alanine, Glycine). These are released as heat breaks down proteins.
- Three Key Stages of the Maillard Reaction:
- Condensation: The reducing sugar’s carbonyl group reacts with the amino acid’s amino group to form an unstable intermediate known as a Schiff base. Water is released.
- Rearrangement: The Schiff base rearranges into a more stable compound called the Amadori product (or Heyns product for ketose sugars like fructose). These products are key intermediates that signal the start of browning and are odorless.
- Degradation and Polymerization: The Amadori products break down further through complex pathways (fragmentation, cyclization) to form two major groups of flavor and color compounds:
- Intermediate Flavor Compounds: Aldehydes and alpha-dicarbonyls.
- End Products (Melanoidins): Large, brown, nitrogenous polymers that contribute to the coffee’s color, body, and overall bitterness.
Page 2/3: Flavor Compounds from Maillard Chemistry
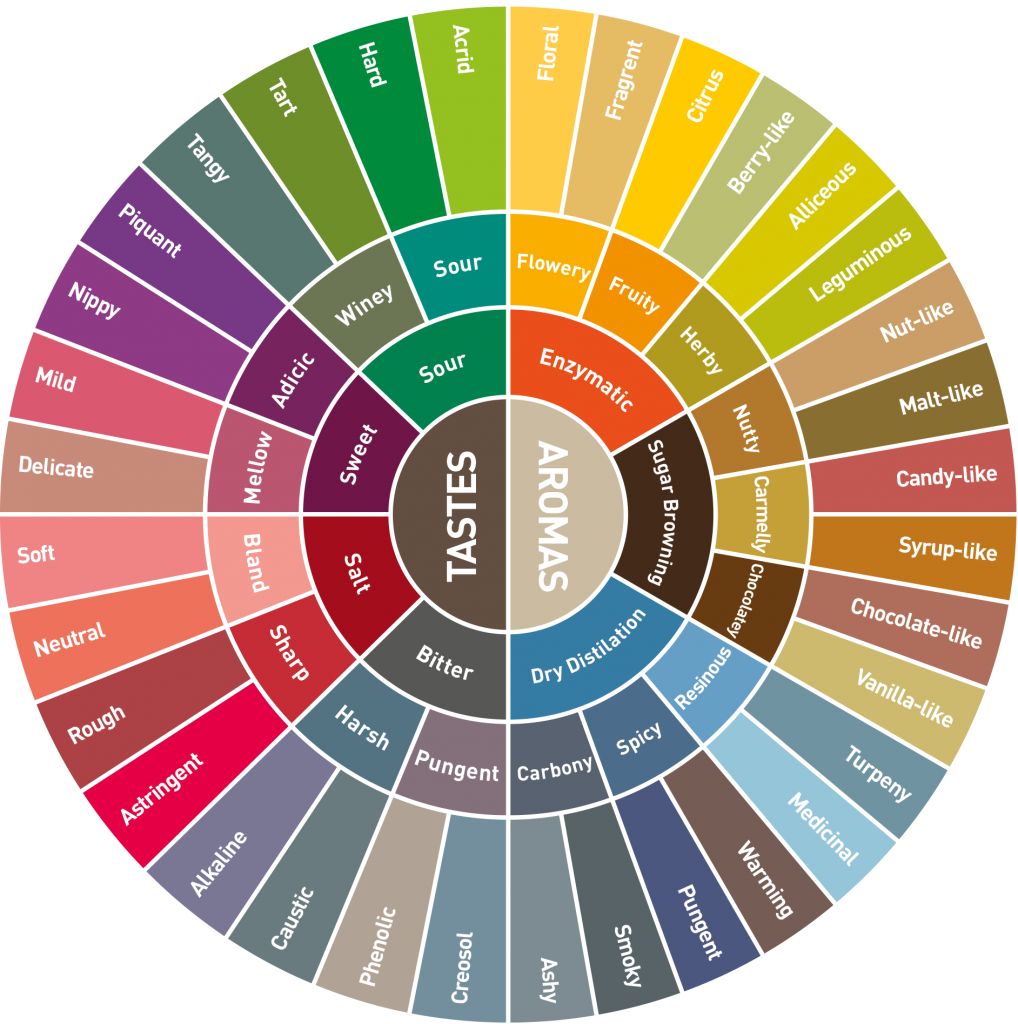
- Hundreds of Volatile Compounds: The final stage of the Maillard reaction creates hundreds of volatile organic compounds (VOCs), which give coffee its characteristic aroma. These compounds are grouped by their chemical structure and associated flavor notes:
- Pyrazines (Nutty/Roasty): Formed from the reaction and condensation of amino acid and sugar fragments. They contribute notes like nutty, earthy, cereal, and bread-like. They increase significantly as the roast darkens.
- Pyrroles & Furans (Caramel/Sweet): Also formed from sugars. They contribute notes like caramel, dark chocolate, and cooked sugar. They are crucial for the perceived sweetness and depth.
- Thiophenes & Thiazoles (Sulfur/Meat): Contain sulfur and contribute savory or meaty notes, though generally subtle in coffee.
- Maillard and Kenyan Coffee (The Sweetness Base):
- Kenyan beans, due to high-altitude maturation, contain a high concentration of initial reactants: free sugars (sucrose) and a rich profile of amino acids.
- The early-stage Maillard reaction is critical for developing the foundation of sweetness (caramel notes from Furans) that balances the highly complex, bright acidity.
- A properly controlled Maillard phase ensures that the desirable sweet/nutty notes develop fully before the onset of pyrolysis (which introduces bitterness).
- Maillard vs. Caramelization:
- Maillard: Requires both amino acids and reducing sugars. Occurs at slightly lower temperatures (140–165^circtext{C}). Produces nitrogen-containing compounds (pyrazines).
- Caramelization: Requires only sugars. Occurs at higher temperatures (>165^circtext{C}). Produces pure sugar breakdown products.
Page 3/3: Ideal Maillard Temperatures and Practical Application
- Roasting Stages and Temperature Zones: Coffee roasting is divided into four main thermal zones:
- Drying Phase (text{Ambient} to approx 100^circtext{C}): Primarily water removal.
- Yellowing Phase (approx 100^circtext{C} to First Crack): The Maillard Zone.
- First Crack/Development (text{First Crack} to text{Drop}): Caramelization, Strecker, Pyrolysis.
- Cooling.
- Ideal Maillard Temps (mathbf{140–165^circtext{C}}):
- This temperature window is the ‘sweet spot’ where the chemical kinetics of the condensation and rearrangement steps are fast enough to progress but slow enough to avoid scorching the sugars.
- Start (approx 140^circtext{C}): The bean turns from green to yellow, emitting aromas of toast/hay (mostly volatiles being driven off).
- Peak (approx 150–165^circtext{C}): The bean turns a pale cinnamon color. Complex Amadori products are formed, and the creation of Pyrazines and Furans accelerates. This phase is also known as the Color Change Phase.
- Roast Profile Strategy: The speed (Rate of Rise, ROR) through this phase is critical. A slower ROR (longer time) in the 150–165^circtext{C} range is thought to encourage more complex Maillard products, increasing perceived sweetness and body. A fast ROR (short time) preserves more acidity but can result in underdeveloped sweetness.
- Practical: Roast to Different Maillard Stages
- Control Roast (Standard): Target 3-4 minutes in the Maillard zone (Yellow to First Crack).
- Underdeveloped Roast (Fast Maillard): Rush through the Maillard zone (e.g., 2 minutes).
- Overdeveloped Roast (Slow Maillard): Drag the Maillard zone out (e.g., 6-7 minutes).
- Sensory Outcome:
- Underdeveloped: Tastes grassy/vegetal, thin, and perhaps overly acidic due to lack of sweetness development.
- Control/Optimal: Balanced sweetness, body, and acidity. Rich nutty/caramel notes.
- Overdeveloped: Tastes roasty, dull, and potentially baked, losing the unique Kenyan acidity.
Day 12: Caramelization & Sugar Breakdown – Notes
Topic 1: Fructose vs. Sucrose Caramelization
Page 1/3: The Process of Caramelization
- Caramelization Definition: Thermal decomposition of carbohydrates (sugars) in the absence of amino acids. It occurs at higher temperatures than the Maillard reaction. This reaction is solely responsible for creating the classic “caramel” and “toffee” notes.
- Reactants in Coffee: The main sugar is sucrose (a disaccharide), which breaks down into the monosaccharides glucose and fructose.
- Chemical Steps (Simplified):
- Inversion/Hydrolysis: Sucrose is broken down by heat/acids into Glucose and Fructose.
- Dehydration: Monosaccharides lose water molecules, forming highly reactive intermediates.
- Fragmentation: Intermediates break down to smaller volatile compounds like diacetyl (butter/butterscotch) and furanones (sweet/cooked fruit).
- Polymerization: Smaller compounds combine to form larger, brown polymers called caramelans (color and bitterness).
- Monosaccharide Melting/Caramelization Points: The temperature required to initiate caramelization depends on the sugar’s structure:
- Fructose: Caramelizes at the lowest temperature (approx 110^circtext{C}) because of its lower melting point and high reactivity.
- Glucose: Caramelizes around 150^circtext{C}.
- Sucrose: Caramelizes around 160^circtext{C}.
Page 2/3: Fructose vs. Sucrose in Coffee Flavor
- The Early Caramelization Role of Fructose:
- Fructose, formed from the breakdown of sucrose, starts to decompose at temperatures significantly lower than the coffee’s peak roast temperature. This means some degree of fructose caramelization and fragmentation occurs early—sometimes even overlapping the late Maillard phase.
- Flavor Contribution: Fructose breakdown products are associated with the desirable fruity, honey, and light caramel notes found in light and medium roasts. Its low caramelization point helps to build initial sweetness and body without requiring excessive heat.
- The Dominant Role of Sucrose (and Glucose):
- Sucrose is the most abundant sugar, and its breakdown during the transition into and past first crack is the primary source of the deeper, darker caramel flavors.
- As the roast progresses into the post-First Crack Development Phase (195–215^circtext{C}), the bulk of the remaining sugars (sucrose and glucose) rapidly caramelize.
- Flavor Contribution: This phase creates the molasses, toffee, and burnt sugar notes of darker roasts. Over-caramelization leads to excessive caramelans, which increase bitterness and decrease overall sweetness.
- How Kenyan Coffees Caramelize Differently:
- High Initial Sugar Content: Kenyan coffees (due to high-altitude slow maturation) typically have higher initial sucrose content compared to low-grade beans. This provides a greater flavor budget for caramelization.
- Acidity Buffer: The exceptionally high concentration of organic acids (Citric, Malic) in Kenyan beans can slightly slow or modify the caramelization reaction kinetics.
- Roasting Strategy: Kenyan coffees are often roasted lighter to preserve the bright acidity. Therefore, roasters intentionally stop the roast before the deep, darker caramelization products fully dominate the flavor. The goal is to maximize the light, clean sweetness from the early fructose and limited sucrose breakdown, allowing the acidity to shine.
Page 3/3: Practical: Taste Light vs. Dark Caramelization Roasts
- Objective: To taste the sensory difference between the flavor compounds created at different stages of sugar breakdown.
- Light Caramelization Roast (Short Development):
- Characteristics: Dropped just past First Crack, with a short Development Time Ratio (DTR) of 15%-20%.
- Chemistry: Fructose breakdown is near completion; Sucrose breakdown is partial. High preservation of volatile acids.
- Sensory: Bright acidity (Citric/Malic), floral/fruity notes, and light, clean honey/candy-like sweetness.
- Dark Caramelization Roast (Longer Development):
- Characteristics: Dropped well into the development phase, possibly near Second Crack, with a high DTR (25%+).
- Chemistry: Near-total destruction of Citric and Malic acids. Massive breakdown of Sucrose and Glucose. High formation of bitter caramelans and quinic acid.
- Sensory: Low acidity, heavy body, dominant notes of dark chocolate, burnt sugar, toffee, and high bitterness.
- Lab Procedure:
- Roast a single batch of Kenyan green coffee to two distinct end points (Light-Medium vs. Medium-Dark).
- Cup the two roasts side-by-side using a standard cupping protocol.
- Identify: The loss of malic/citric acidity and the gain of quinic acid/caramel bitterness.
- Conclusion: The lab illustrates that the high concentration of initial sugars in Kenyan coffee can be manipulated to produce two vastly different—but still high-quality—cup profiles based on the degree of caramelization allowed.
Day 13: Strecker Degradation & Aroma Formation – Notes
Topic 1: Formation of Pyrazines, Furans (Nutty, Caramel Notes)
Page 1/3: Strecker Degradation as a Secondary Reaction
- Definition: Strecker Degradation is a specific chemical reaction that occurs as a consequence of the Maillard reaction. It is the decomposition of an alpha-amino acid by an alpha-dicarbonyl compound (an intermediate of the Maillard reaction).
- The Key Transformation:
- Reactants: alpha-Amino Acid + alpha-Dicarbonyl (e.g., from sugar fragmentation).
- Products: An Aldehyde (called the Strecker Aldehyde) + text{CO}_2 + an Aminoketone.
- Crucial Role of Strecker Aldehydes:
- The aldehydes created in this reaction are highly reactive and are themselves a major group of volatile aroma compounds (VOCs).
- More importantly, these aldehydes and aminoketones rapidly react with other compounds, often through cyclization (forming rings) and condensation (joining together), to create some of the most potent and recognizable coffee aroma molecules: Pyrazines and Furans.
- Aroma Compound Groupings (Review):
- Pyrazines: Formed by the self-condensation of two alpha-aminoketone molecules. Responsible for the nutty, roasty, and earthy notes. They are highly stable and are dominant in the final stages of the roast.
- Furans: Formed from the dehydration and fragmentation of sugars and Strecker aldehydes. Contribute caramel, sweet, buttery, and cooked-fruit notes. They are often the core of the perceived sweetness and depth.
Page 2/3: Strecker, Pyrazines, and the Roast Profile
- Pyrazines as Roast Markers:
- The concentration of pyrazines is directly correlated with the degree of roast. They begin forming actively in the later Maillard phase (160^circtext{C}) and increase substantially through First Crack and into pyrolysis.
- Light Roast: Lower pyrazine concentration, resulting in a cleaner cup with a focus on origin acids and high-toned fruit.
- Dark Roast: High pyrazine concentration, resulting in a dominant “roasty” or “smoky” flavor that often masks the original origin characteristics.
- Furanones and the Sweet Spot:
- Furanones (a type of Furan) are key to the caramelized sweetness. Furaneol is a specific furanone that gives sweet, strawberry-like aromas.
- The balance of furan formation is critical in the Maillard/Development phase. If the roast ends too early, the furan-based sweetness is underdeveloped; if it goes too long, they are degraded, and the sweetness is masked by bitterness.
- Why Kenyan Coffee Has Floral/Fruity Aromas (Specific Precursors):
- High Amino Acid Diversity: Kenyan high-altitude beans, particularly the SL varietals, contain a high concentration of specific, complex amino acids, like Proline, Alanine, and Phenylalanine.
- Precursor-to-Aroma Link: When these specific amino acids undergo Strecker degradation, they produce unique Strecker Aldehydes that lead to highly volatile and specific aroma molecules:
- Aldehydes from Phenylalanine rightarrow Floral notes (e.g., phenylethyl acetate).
- Aldehydes from Leucine/Isoleucine rightarrow Fruity notes.
- High Acidity Preservation: Since Kenyan coffee is almost always roasted light to medium to preserve the high acidity, these delicate, high-toned floral and fruity volatile compounds are preserved. In a dark roast, the sheer volume of nutty/roasty pyrazines and quinic acid bitterness would entirely overpower these floral notes.
Page 3/3: Practical: Aroma Identification Lab
- Objective: To distinguish between the different volatile compounds created by various chemical reactions, with a focus on Strecker degradation products (Pyrazines, Furans, etc.).
- Aroma Standards: Use vials containing key synthetic aroma compounds dissolved in a neutral base:
- Pyrazines (e.g., 2,3,5-Trimethylpyrazine): Roasty, Nutty, Baked Bread (Strecker Product).
- Furanones/Furans (e.g., Furaneol): Caramel, Sweet, Butterscotch (Maillard/Caramelization/Strecker Product).
- Acetic Acid: Vinegar, Pungent (Fermentation/Roasting Volatile).
- Aldehydes (e.g., Phenylacetaldehyde): Floral, Honey-like (Direct Strecker Aldehyde).
- Quinic Acid/Phenols: Harsh, Medicinal, Astringent (Pyrolysis Product).
- Cupping Correlation:
- Blindly taste and evaluate three different Kenyan roast profiles (Light, Medium, Dark).
- Correlate the aromas smelled in the standards to the aromas perceived in the brewed coffee.
- Expected Link: The Light Roast will show a higher sensory link to the Floral/Fruity Aldehyde standards (preserved Strecker products). The Dark Roast will show a higher link to the Pyrazine and Quinic Acid standards (pyrolysis and maximum Strecker/Maillard polymerization).
- Conclusion: The lab reinforces that the unique Kenyan flavor is a direct result of preserving the more delicate, high-toned volatiles produced by specific Strecker degradation pathways before they are overwhelmed by the heavier pyrazines of darker roasting.
Day 14: First Crack Chemistry – Notes
Topic 1: Water Vaporization and Cellulose Breakdown
Page 1/3: Pre-Crack—The Endothermic Phase
- First Crack Definition: The first audible “popping” sound during coffee roasting, analogous to popcorn popping. It signals a major shift in the physical and chemical state of the bean and marks the end of the Maillard phase.
- Precursors to First Crack:
- Drying Phase (End of text{Drying} to approx 100^circtext{C}): Free surface water is removed.
- Yellowing Phase (approx 100^circtext{C} to text{First Crack}): Water still bound within the cell structure is being converted to steam. This process is highly endothermic (heat-absorbing), requiring the roaster to continuously apply heat to maintain a rising text{Rate of Rise (ROR)}.
- The Critical Role of Water Vaporization (mathbf{approx 180^circtext{C}}):
- As the internal bean temperature approaches and exceeds the boiling point of water (text{>100}^circtext{C}), the water trapped inside the dense cellulose cell structure converts into high-pressure steam (water vapor).
- This steam cannot easily escape the bean due to the low permeability of the cell walls and the central crease (sulcus). The internal pressure begins to build rapidly.
- Start of Cellulose Breakdown:
- Cellulose: The rigid polymer that forms the bulk of the bean’s cell walls. It provides the bean’s structure.
- Just before and during First Crack, the high internal temperature begins to cause the initial pyrolysis (thermal decomposition) of the hemicellulose and the less-ordered (amorphous) regions of the cellulose matrix. This is the first significant change in the bean’s solid structure.
- Products: This decomposition releases water, text{CO}_2, text{CO}, and other volatile gases, further adding to the internal pressure.
Page 2/3: The Physics of the Crack and Bean Expansion
- The Mechanism of First Crack:
- The continuous buildup of internal pressure (primarily steam/water vapor, but also text{CO}_2 and other gases from early pyrolysis) eventually exceeds the tensile strength of the bean’s rigid cellulose cell walls.
- This pressure is relieved by the fracture or rupture of the bean structure, resulting in the distinct pop or snap sound.
- Sensory/Visual Indicators: The bean rapidly expands in volume (swelling), the central crease begins to open, and the bean surface often becomes slightly smoother as the structure distorts.
- The Exothermic Shift (Post-First Crack):
- First Crack often marks the transition from a highly endothermic (heat-absorbing) phase to a weakly exothermic (heat-releasing) phase.
- The pyrolytic reactions (decomposition of cellulose, sugars, acids) that break the bean structure are highly energy-releasing. The heat generated by these chemical reactions partially offsets the heat that must be supplied by the roaster.
- Roaster Action: The roaster must drastically reduce the applied heat immediately after First Crack to prevent the text{ROR} from accelerating uncontrollably, which would “flick” the roast and lead to a burnt exterior/underdeveloped interior.
- Pressure Buildup in Kenyan Dense Beans:
- High Density: Kenyan beans are typically characterized as Strictly High Grown (SHG) and are extremely dense due to the cool, slow maturation at high altitude.
- Impact on First Crack: A denser bean structure means:
- Less Porosity: The internal structure is tighter, providing more resistance for steam/gas to escape.
- Higher Tensile Strength: The cell walls are potentially stronger.
- Result: The internal pressure required to fracture the bean is higher than in a low-density bean. This often means:
- Higher First Crack Temperature: First Crack may occur at a slightly higher temperature (due to the need for more energy/pressure).
- More Energetic Crack: The pop can be louder and more pronounced.
- Faster text{ROR} Drop: The exothermic reaction can be more pronounced, requiring quicker heat reduction by the operator.
Page 3/3: Practical: Record First Crack Temperatures
- Objective: To precisely identify the temperature marker for the chemical and physical shift of First Crack and use it to manage the development phase.
- Equipment: Roaster with a digital temperature logger (e.g., thermocouples logging via software like Artisan/Cropster).
- Procedure:
- Roast a batch of Kenyan green coffee, carefully logging the bean temperature.
- Use the logged data to identify the exact text{Bean Temperature at First Crack (BT_FC)}.
- Repeat with a lower-density bean (e.g., a low-altitude Central American).
- Compare the text{BT_FC} and the text{ROR} drop immediately following the crack.
- Expected Results and Roaster Strategy:
- Kenyan BT_FC: Expected to be slightly higher than the low-density bean (e.g., 198-205^circtext{C}) due to the high density and pressure requirement.
- ROR Management: The roaster must reduce heat aggressively immediately after text{BT_FC} to establish a controlled Development Time Ratio (DTR). For a high-density Kenyan, failure to drop the heat immediately can result in an uncontrollable text{ROR} spike and a baked flavor.
- Conclusion: text{First Crack} is not just an audible marker; it is the critical point where the bean transitions from a structure primarily composed of intact cellulose and sugars to a porous structure undergoing rapid pyrolytic breakdown. Its exact temperature is essential for managing the heat and ensuring flavor development without burning.
Day 15: Roast Development & Pyrolysis – Notes
Topic 1: How text{CO}_2 Forms During Roasting
Page 1/3: Pyrolysis – The Destructive Phase
- Pyrolysis Definition: The chemical decomposition of organic materials (in this case, coffee compounds) caused by high temperatures in the absence of oxygen. In coffee, it occurs most significantly after First Crack and is the primary source of the deep brown color, bitter notes, and high gas pressure.
- The Main Reactants: The large, complex molecules that survived the Maillard/Caramelization phases:
- Carbohydrates: Remaining cellulose, hemicellulose, and polymerized sugars (melanoidins, caramelans).
- Acids: Chlorogenic Acids (CGAs) and organic acids.
- Trigonelline: An alkaloid (not caffeine) that is a major contributor to bitterness.
- text{CO}_2 Formation Mechanisms (Primary Source):
- Carbohydrate Pyrolysis: High heat breaks the long chains of cellulose and hemicellulose. These reactions strip oxygen and hydrogen atoms from the molecules, forming small volatile fragments, including a large volume of text{CO}_2.
- Thermal Decomposition of Organic Acids: Remaining acids (especially Chlorogenic Acids) undergo heat-driven decomposition (text{decarboxylation}). This process specifically removes a carboxyl group (-text{COOH}) from the molecule, which is released as a molecule of carbon dioxide (text{CO}_2).
text{R-COOH} xrightarrow{text{Heat}} text{R-H} + text{CO}_2
- Impact of text{CO}_2 Formation:
- The rapid formation and subsequent pressure buildup of text{CO}_2 is the primary cause of the Second Crack (a louder, sharper crack than the first) in darker roasts, as the remaining internal structure fractures again.
- A portion of this text{CO}_2 remains trapped in the bean’s now-porous matrix, leading to the phenomenon of degassing (Day 17).
Page 2/3: Balancing Development Time for Kenyan Beans
- Development Time (DT): The time period from the start of First Crack to the end of the roast (when the beans are dropped/cooled).
- Development Time Ratio (DTR): The Development Time expressed as a percentage of the Total Roast Time. (e.g., 10 minute roast, 2 minutes DT rightarrow 20%text{ DTR}).
- Goal of DT/DTR: To ensure the chemical reactions (pyrolysis, caramelization) are complete enough to fully develop the bean’s internal flavor structure without destroying the desirable high-toned compounds.
- Strategy for Kenyan Beans (Light/Medium Roast):
- Challenge: Kenyan beans are valued for their bright, complex acidity (Citric, Malic, Phosphoric). These acids are volatile and are rapidly destroyed during pyrolysis/long DT.
- The Balancing Act: The roaster needs just enough development to:
- Convert the grassy/vegetal flavor of the under-roasted core.
- Create some text{CO}_2 for freshness/crema (espresso).
- Develop the base sweetness (Maillard/Caramelization).
- Typical Target: A relatively short DT (approx 1:30 – 2:30 minutes) leading to a low DTR (e.g., 18% – 22%). This quickly moves the bean past the grassy stage but stops before the desirable acids are destroyed and the harsh quinic acid/pyrazines dominate.
- Underdeveloped vs. Overdeveloped Kenyan:
- Underdeveloped (Too Short DT): Grassy, vegetal, hollow flavor; acidity is sharp but one-dimensional.
- Overdeveloped (Too Long DT): Flat, dull, “baked” flavor; bright acidity is gone, replaced by a dull bitterness from excessive CGA and Trigonelline decomposition.
Page 3/3: Practical: Measure Roast Degree with Agtron
- Objective: To quantify the degree of roast precisely and link the measurement to the chemical state (amount of pyrolysis/pyrolytic text{CO}_2 formation).
- Agtron Gourmet Scale: A standardized, numerical scale (0-100) used to measure the color (roast degree) of whole beans and ground coffee, where 100 is green and 0 is charcoal. Specialty coffee often targets 55-70.
- Instrumentation: An Agtron color analyzer (a specialized spectrophotometer) measures the light reflectance of the coffee.
- Procedure:
- Roast a batch of Kenyan coffee, dropping samples at 30-second intervals post-First Crack.
- Grind and measure the color of each sample with the Agtron.
- Cup the samples blind.
- Correlation: Find the text{Agtron} number that corresponds to the desired Kenyan flavor profile (high acidity, balanced sweetness). This number is the target for future roasts.
- Chemical Link: The text{Agtron} number directly correlates with the amount of melanoidin (brown pigment) and caramelan formation, which are the main end products of Maillard/Caramelization/Pyrolysis. A low text{Agtron} number (darker roast) indicates a higher degree of pyrolysis and therefore a higher concentration of text{CO}_2-forming reactions.
- Conclusion: By using the Agtron, the roaster can move beyond subjective visual inspection and use a precise, repeatable scientific measurement to balance the minimal necessary pyrolysis against the desired preservation of the Kenyan coffee’s delicate, high-toned flavor compounds.
Day 16: Quenching & Roast Stabilization – Notes
Topic 1: Why Fast Cooling Preserves Acidity
Page 1/3: The Quenching Principle
- Quenching Definition: The process of rapidly stopping the chemical reactions in the coffee bean by applying external cooling immediately after the roast is dropped from the drum. This is typically done using high-velocity forced air or a fine mist of water (water quenching).
- The Chemical Momentum: When the bean is dropped, its internal temperature is very high (approx 200-215^circtext{C}). Even when removed from the heat source, the core of the bean retains a significant amount of latent heat.
- The Hazard of Slow Cooling:
- If the beans are cooled too slowly (e.g., leaving them to air cool without forced air), the chemical reactions—especially pyrolysis (the thermal decomposition of acids and sugars) and the formation of quinic acid—continue even outside the drum.
- This is known as coasting or baking and is highly detrimental to specialty coffee flavor.
- The Role of Fast Cooling (Quenching):
- Fast cooling rapidly brings the bean temperature down to a safe point (e.g., below 50^circtext{C} in under 4 minutes) where chemical reaction rates are slowed to near-zero.
- Preserving Desirable Volatiles:
- The primary chemical benefit of rapid quenching is the preservation of volatile organic acids (Citric, Malic) and delicate, high-toned aroma compounds (Floral Aldehydes).
- Slowing the rate of chemical decomposition prevents the conversion of these desirable compounds into less desirable, harsher, or heavier compounds (e.g., sweet Citric Acid rightarrow harsh Quinic Acid).
Page 2/3: The Chemical Cascade of Slow Cooling
- Targeted Decomposition (What we don’t want):
- Organic Acid Loss: Slow cooling provides the extra thermal energy required to continue breaking down Malic and Citric acids into simpler, often flavorless, compounds or undesirable ones like acetic acid. This causes a noticeable loss of brightness and clarity.
- Increased Quinic Acid: The thermal decomposition of Chlorogenic Acids (CGAs) continues, leading to an unwanted increase in the concentration of bitter and astringent quinic acid. This results in a dry and harsh finish (the “baked” taste).
- Aroma Loss: Volatile aroma compounds (VOCs) that were successfully formed during the Maillard/Strecker reactions continue to vaporize and escape from the bean during the extended cooling period, leading to a muted, flat aroma profile.
- Kenyan Roast Profiles: Fast vs. Slow Cooling:
- Kenyan Goal: Maximize fruit, floral, and bright acidity.
- Fast Cooling (Standard Kenyan Profile): Essential. By stopping the chemistry quickly, the vibrant, high-toned complexity is locked into the bean structure, resulting in a clean, intensely bright, and dynamic cup.
- Slow Cooling (Defective Kenyan Profile): The result is a profile that is dull, heavy, and baked-tasting, losing all of the expensive, high-altitude flavor characteristics. The flavor profile will shift towards a more typical “medium-dark” profile, regardless of the drop temperature, because the post-roast reactions overdevelop the bean.
Page 3/3: Practical: Test Different Quenching Methods
- Objective: To demonstrate the sensory impact of the cooling rate on the final cup chemistry.
- Experimental Design: Roast two identical batches of Kenyan coffee to the exact same drop temperature and time, then apply two different cooling methods:
- Fast Quench (Control): High-speed forced air cooling (target: below 50^circtext{C} in <3 minutes).
- Slow Quench (Test): Low-speed or ambient cooling only (target: 6-10 minutes to cool).
- Analysis and Measurement:
- Weight Loss: Weigh the beans immediately after cooling. The Slow Quench sample will likely show a slightly higher weight loss (due to further evaporation/gas release).
- Cupping: Blindly cup the two samples.
- Sensory Outcome: The Fast Quench sample should exhibit the classic Kenyan attributes: high, complex acidity, floral aroma, and clean sweetness. The Slow Quench sample will be noticeably flatter, less aromatic, and have a harsh, drying aftertaste (indicative of increased quinic acid and destruction of desirable volatiles).
- Roaster Takeaway: Quenching is the final critical control point in the roast profile. It’s the step that stabilizes the achieved chemistry. For high-acidity, high-value coffees like Kenyan, rapid quenching is non-negotiable for preserving origin flavor.
Day 17: text{CO}_2 Degassing & Freshness – Notes
Topic 1: How Kenyan High-Density Beans Degas Slower
Page 1/3: The Science of Degassing
- Degassing Definition: The slow release of trapped gases, primarily carbon dioxide (text{CO}_2), from the roasted coffee bean over time.
- text{CO}_2 Source: text{CO}_2 is a major by-product of Pyrolysis (Day 15) and other thermal decomposition reactions that occur post-First Crack. A significant portion of this gas remains trapped in the bean’s microstructure.
- Role in Brewing: text{CO}_2 is essential for freshness (it acts as a natural preservative by displacing oxygen), but too much text{CO}_2 interferes with optimal extraction:
- Bloom (Filter): text{CO}_2 rapid release causes the grounds to “bloom,” helping to evenly saturate the coffee.
- Extraction Interference: During the main brew, the continuous release of text{CO}_2 creates bubbles that repel water from the coffee particles. This prevents even wetting and can lead to channeling (in espresso) or under-extraction (in filter), resulting in a sour/sharp taste.
- Factors Affecting Degassing Rate:
- Roast Degree: Darker roasts have more advanced pyrolysis, creating more text{CO}_2, but their structure is more porous (more fractured cell walls), so they degas faster.
- Bean Density: Low-density beans are more porous and degas faster.
- Grind Size: Ground coffee degasses extremely fast (minutes to hours) due to the massive increase in surface area.
Page 2/3: The Kenyan Density Factor
- Kenyan Bean Structure: Kenyan coffee beans are known for their high density (Strictly Hard Bean) due to high-altitude, slow maturation. They are almost always roasted to a light or medium degree to preserve acidity.
- Impact of Density and Roast Level on Degassing:
- Lower Porosity: The high-density structure provides more resistance to gas escape. The cell walls are strong, and the internal pathways for text{CO}_2 release are restricted.
- Lighter Roast: Since light roasts undergo less pyrolysis, their overall cell structure is less fractured, keeping more text{CO}_2 trapped inside.
- The Slower Degassing Result: The combination of high density and light roast means Kenyan coffees degas significantly slower than darker or lower-density beans.
- They require a longer resting (aging) time post-roast to allow sufficient text{CO}_2 to escape, which minimizes extraction interference during brewing.
- Practical Consequence for Roasters: A Kenyan coffee brewed too soon (e.g., 1-3 days post-roast) will often taste harsh, sour, and unevenly extracted, not because the roast is bad, but because the high residual text{CO}_2 is preventing optimal water contact.
Page 3/3: Optimal Resting Time for Brewing & Practical
- Optimal Resting Time for Brewing (Aging): The necessary rest time is a function of density and roast level, with the goal being to lower the residual text{CO}_2 to a level that allows for even extraction.
- Dark Roast/Low Density: May be ready in 2-5 days.
- Light Roast/High Density (Kenyan): Typically requires 7-14 days minimum, and often tastes best between 10 and 21 days post-roast. The extended rest allows the complex acids and sweetness to fully “settle” and be extracted cleanly.
- Practical: Measure text{CO}_2 Release using Water Displacement
- Objective: To quantify the text{CO}_2 release rate over time.
- Method (Simplified Manometry/Water Displacement):
- Place a known weight of freshly roasted Kenyan coffee (Day 1 post-roast) into a sealed chamber connected to a water displacement system (e.g., a simple inverted, calibrated beaker submerged in water).
- Measure the volume of water displaced by the text{CO}_2 gas released over a fixed period (e.g., 24 hours).
- Repeat the measurement with the same coffee at Day 5, Day 10, and Day 15 post-roast.
- Expected Results: The volume of text{CO}_2 released per 24-hour period will significantly decrease over time, proving the slow degassing rate. The greatest volume of gas will be collected on Day 1 and Day 2.
- Conclusion: This lab provides a physical, quantitative proof of the necessity of resting, demonstrating that degassing is a chemical decay curve that must be managed to achieve optimal solubility and flavor clarity in high-density Kenyan beans.
Day 18: Grinding & Particle Size Chemistry – Notes
Topic 1: Surface Area and Solubility
Page 1/3: The Chemistry of the Coffee Particle
- Grinding Definition: The mechanical breakdown of roasted coffee beans into smaller particles. This is the last process before brewing and is the single most important variable a barista controls for extraction.
- Surface Area and Extraction Kinetics:
- Solubility: The rate at which the soluble compounds (acids, sugars, melanoidins, etc.) dissolve into the hot brewing water.
- Surface Area Relationship: The total surface area of the coffee particles in contact with the water directly determines the rate of extraction.
- Finer Grind: rightarrow Massive increase in total surface area rightarrow Faster extraction rate.
- Coarser Grind: rightarrow Lower total surface area rightarrow Slower extraction rate.
- The Layer Model of Extraction: Extraction occurs in three main stages regardless of grind size, but the rate of transition between stages is controlled by surface area:
- Fast Extraction (Surface): Rapid dissolution of compounds easily accessible on the surface (Acids, small molecules, salts).
- Moderate Extraction (Matrix): Slow dissolution of compounds from deeper within the porous matrix.
- Slow Extraction (Undesirables): Dissolution of less soluble, often bitter compounds (phenols, cellulose breakdown products) from the bean core.
- Grind Consistency (Homogeneity): The most crucial aspect. An ideal grind size is uniform. Poor consistency creates a mix of:
- Fines (too small): Over-extract rapidly (bitter/astringent).
- Boulders (too large): Under-extract rapidly (sour/salty).
Page 2/3: Kenyan Grind Settings for Espresso vs. Pour-Over
- The Grind/Brew Method Relationship: Each brewing method dictates a necessary grind size based on two factors: Brew Time and Water Pressure.
- Espresso (High Pressure, Short Time):
- Required Grind: Very Fine (like flour/powder).
- Chemistry: The high water pressure (up to 9 bar) and short contact time (approx 25-35 seconds) necessitate a very high surface area to achieve optimal extraction yield (18%-22%).
- Kenyan Specifics: Kenyan beans are dense and often roasted light (hard). When ground, they tend to produce a higher percentage of undesirable fines (small particles) than softer beans. This requires careful grinder selection and often a slightly coarser setting than expected to prevent the portafilter from choking (blocking the flow).
- Pour-Over (Low Pressure, Medium Time):
- Required Grind: Medium-Fine (like table salt).
- Chemistry: A longer contact time (approx 2:30 – 4:00 minutes) at atmospheric pressure requires a medium particle size. A finer grind would clog the filter and lead to excessive contact time and bitterness.
- Kenyan Specifics: For a light-roasted Kenyan, a slightly finer pour-over grind may be used than for other coffees. This is to help increase the extraction rate and achieve the desired yield, counteracting the effects of the bean’s high density and slow solubility. High heat (text{>94}^circtext{C}) is also used to aid solubility.
Page 3/3: Practical: Sieve Analysis of Different Grinds
- Objective: To quantitatively measure the particle size distribution (PSD) of a grind sample.
- Instrumentation: A stack of standardized sieves (sifters) with specific mesh sizes (measured in microns, mutext{m}).
- Procedure:
- Grind a 50text{g} sample of roasted Kenyan coffee at an Espresso setting and a second sample at a Pour-Over setting.
- Pour each sample through the stacked sieves.
- Mechanically shake or vibrate the sieves for a fixed time.
- Weigh the mass of coffee retained on each sieve (e.g., text{>850}mutext{m}, 600-850mutext{m}, text{<200}mutext{m} (fines)).
- Analysis and Conclusion:
- Target: The Pour-Over grind should have the majority of its mass concentrated in a middle range (e.g., 600-850mutext{m}), while the Espresso grind should be concentrated in a lower range.
- Fines Quantification: The lab visually and quantitatively identifies the percentage of fines (small particles that cause bitterness/clogging). This highlights the challenge of grinding dense, light-roasted Kenyan beans and informs the optimal grinder choice and brewing strategy.
Day 19: Water Chemistry & Extraction – Notes
Topic 1: Ideal TDS, text{pH}, and Mineral Content
Page 1/3: Water as the Solvent in Coffee
- Water’s Role: Water is the single most abundant ingredient in the final cup (text{>98}%) and is the solvent that carries the extracted coffee compounds. Its chemistry is critical.
- Total Dissolved Solids (TDS):
- Definition: The measure of all inorganic (minerals, salts) and organic substances dissolved in the water, expressed in parts per million (text{ppm}).
- SCA Recommendation: 125-175text{ ppm} (ideal is 150text{ ppm}).
- Impact: Too Low text{TDS} (“Soft Water”) rightarrow The water lacks extraction power, leading to flat, under-extracted coffee. Too High text{TDS} (“Hard Water”) rightarrow Can lead to over-extraction of bitter compounds and scale buildup in equipment.
- Water text{pH} (Acidity/Alkalinity):
- SCA Recommendation: Near-neutral text{pH} of 6.5-7.5.
- Impact: Water text{pH} affects the final perceived acidity of the coffee. Acidic water (text{pH <6}) can result in a coffee that tastes overly sour/sharp. Alkaline water (text{pH >7.5}) neutralizes the coffee’s desirable organic acids (Citric, Malic), leading to a flat, dull-tasting cup.
Page 2/3: The Key Minerals in Extraction
- Carbonate Hardness (Alkalinity):
- Definition: The measure of bicarbonate and carbonate ions (e.g., text{HCO}_3^-) in the water. Measured in text{ppm} of text{CaCO}_3.
- SCA Recommendation: 40-75text{ ppm}.
- Role (Buffering): Bicarbonate ions act as a buffer, neutralizing the coffee’s desirable organic acids.
- Too Low Alkalinity: Coffee acids are not buffered, leading to an overwhelmingly sharp, sour, and unbalanced taste.
- Too High Alkalinity: Coffee acids are over-neutralized, leading to a flat, dull, and chalky taste where the characteristic Kenyan brightness is lost.
- General Hardness (Calcium and Magnesium):
- Definition: The measure of divalent metal ions, primarily text{Ca}^{2+} (Calcium) and text{Mg}^{2+} (Magnesium).
- SCA Recommendation (Ca Hardness): 50-175text{ ppm}.
- Role (Extraction): text{Mg}^{2+} and text{Ca}^{2+} are highly efficient at bonding with and extracting flavor compounds from the coffee grounds.
- Magnesium (text{Mg}^{2+}): Especially good at extracting the complex, desirable fruity and floral compounds (like those in Kenyan coffee), often enhancing brightness.
- Calcium (text{Ca}^{2+}): Good for general extraction and contributing to body, but excessive text{Ca}^{2+} (high hardness) often dulls the flavor and causes scale.
Page 3/3: How Kenyan Water Hardness Affects Extraction & Practical
- Kenyan Water Profile (Generalization): Water in many high-altitude, volcanic regions tends to have a complex mineral profile. Depending on the source, it can be relatively soft to moderately hard.
- The Kenyan text{Mg}^{2+} Hypothesis: Because Kenyan coffee is sought after for its bright, fruity, and floral notes, water with a relatively high ratio of Magnesium to Calcium is often favored. Magnesium preferentially extracts the desired acid-based flavor compounds that define the Kenyan profile.
- The Alkalinity Risk: Using local water with high alkalinity is the primary risk when brewing Kenyan coffee. The high bicarbonate level will neutralize the high concentration of Citric/Malic/Phosphoric acids inherent in the bean, destroying the coffee’s most valuable attribute: its vibrant acidity.
- Practical: Brew with Different Water Recipes
- Objective: To taste the sensory impact of water chemistry on a high-acidity coffee.
- Recipes:
- Ideal SCA Water (Control): approx 150text{ TDS}, 50text{ Alkalinity}, high text{Mg}^{2+}/text{Ca}^{2+} ratio.
- Zero TDS Water (Flat): Deionized or distilled water.
- High Alkalinity Water (Dull): Water with 150+text{ ppm} alkalinity.
- Sensory Outcome:
- Zero TDS: Coffee will taste sour (under-extracted/harsh) because the water has no extraction power.
- High Alkalinity: Coffee will taste flat and dull—the characteristic Kenyan acidity will be missing.
- Ideal SCA: The coffee will be vibrant, sweet, and balanced, with its full-spectrum of acidity present.
- Conclusion: Water chemistry is a chemical lever for the flavor. Adjusting the water (especially alkalinity and text{Mg}^{2+}) is necessary to unlock the full potential of high-quality, high-acidity Kenyan coffee.
Day 20: Solubility & Extraction Yield – Notes
Topic 1: Ideal Extraction Range (18–22%)
Page 1/3: Principles of Solubility and Extraction
- Solubility and Extraction:
- Solubility: The maximum amount of a substance (coffee compounds) that can dissolve in a solvent (water) at a given temperature.
- Extraction: The process of dissolving these soluble compounds from the coffee grounds during brewing.
- Extraction Yield (EY): The percentage of the initial dry coffee mass that was successfully dissolved into the final beverage.
- text{EY} = (text{Mass of Solubles in Brew} / text{Mass of Dry Coffee Used}) times 100
- The Sequential Extraction Curve (The Three Phases): As water flows through the coffee, it extracts compounds in a predictable sequence:
- Phase 1 (Fast, Early): Acids, Salts, and highly volatile compounds. (If you stop here, the coffee is sour and salty—under-extracted).
- Phase 2 (Moderate, Middle): Sugars, Melanoidins, and deeper-seated Maillard products. (The ideal flavor balance).
- Phase 3 (Slow, Late): Less soluble, heavy compounds, cellulose fragments, and harsh phenols. (If you extract too far, the coffee is bitter and astringent—over-extracted).
- The “Ideal” Range: 18%-22% EY:
- Empirically determined by research (The text{Lockhart} E.K. Brew Control Chart). Within this range, the most desirable balance of Acids (text{sour/sweet}), Sugars (text{sweet}), and Bitterness (text{bitter/body}) is achieved, avoiding the extreme flavors of under- and over-extraction.
Page 2/3: Measuring the Strength: Total Dissolved Solids (TDS)
- Total Dissolved Solids (TDS): The measure of the concentration of coffee solubles in the final beverage.
- SCA Recommendation: 1.15%-1.35% text{TDS} (i.e., 11,500 – 13,500text{ ppm}).
- Relationship between EY and TDS:
- text{EY} is the measure of efficiency (how much was dissolved from the grounds).
- text{TDS} is the measure of concentration (how strong the final cup is).
- They are connected by the Brew Ratio (Coffee Mass : Water Mass).
- text{EY} = (text{TDS} times text{Final Brew Mass}) / text{Dry Coffee Mass}
- How Kenyan Coffee Extracts Differently (Higher Yield Potential):
- High Solubility Potential: Kenyan high-altitude beans, with their high concentration of initial precursors (sugars, amino acids, acids), typically contain a greater percentage of desirable soluble matter.
- Higher Yield Target: Because of their inherent complexity and high-quality precursors, it is often possible—and desirable—to push the extraction of Kenyan coffees slightly higher than the 22% mark (e.g., 23%-24%) while still maintaining a balanced, sweet flavor. This is a testament to the high quality and lack of defects in the green bean chemistry.
- Brewing Implications: The density and hardness of the Kenyan bean means achieving this high yield often requires a finer grind, higher water temperature, and/or more agitation than other coffees.
Page 3/3: Practical: Measure TDS with a Refractometer
- Objective: To precisely measure the TDS and calculate the Extraction Yield (EY) to ensure the brew falls within the optimal 18%-22% range (or the target Kenyan high yield).
- Instrumentation: A coffee refractometer. This device measures the refraction (bending) of light as it passes through the brewed coffee. The more dissolved solids present, the more the light bends. The device converts this measurement to a text{TDS} percentage.
- Procedure:
- Brew a cup of Kenyan coffee using a controlled recipe (e.g., text{V60}, 15text{g} coffee, 250text{g} water).
- Cool a few drops of the final brew and place them on the refractometer prism.
- Read the text{TDS} (e.g., 1.30%).
- Calculate EY: Use the brew ratio formula: text{EY} = (1.30 times 250) / 15 approx 21.67%.
- Troubleshooting: If the result is below 18% (under-extracted/sour), the solution is to grind finer for the next brew. If above 22% (over-extracted/bitter), the solution is to grind coarser.
- Conclusion: The refractometer is the final scientific tool in the coffee chain. It allows the brewer to move beyond subjective taste to make data-driven adjustments (usually to grind size) that ensure the chemical potential locked into the perfectly roasted Kenyan bean is fully realized in the final cup.
Day 21: Final Sensory Evaluation – Notes
Topic 1: Linking Chemical Changes to Flavor
Page 1/3: Synthesis: Chemistry to Sensory
- The Final Objective: To connect the theoretical chemistry learned over the course (Days 8-20) to the actual sensory experience (cupping).
- Chemistry rightarrow Sensory Links:
- Acid Breakdown rightarrow Acidity:
- Preserved: High concentration of Citric, Malic, and Phosphoric acids (light roast, fast cooling) rightarrow Vibrant, bright, complex, clean acidity (lemon, apple, cola notes).
- Degraded: Breakdown of CGAs and desirable acids (dark roast, slow cooling) rightarrow Dull, harsh, bitter/astringent acidity (from Quinic acid).
- Maillard Reaction rightarrow Sweetness/Body:
- Optimal: Balanced formation of Furans and Pyrroles rightarrow Caramel, nutty, chocolate sweetness, with increased body (from Melanoidins).
- Underdeveloped: Insufficient Maillard rightarrow Grassy, vegetal, muted flavor.
- Strecker Degradation rightarrow Aroma:
- Preserved: Specific text{Strecker} aldehydes from high-quality amino acids (light roast) rightarrow Floral, fruity, and winey aromas.
- Dominant: High concentration of Pyrazines (dark roast) rightarrow Roasty, smoky, burnt aromas that overpower origin notes.
- text{CO}_2 Degassing rightarrow Clarity:
- Insufficient Rest: High residual text{CO}_2 rightarrow Sour, sharp, and unevenly extracted taste due to water channeling.
- Optimal Rest: Low residual text{CO}_2 rightarrow Clean, clear, and balanced flavor.
- Acid Breakdown rightarrow Acidity:
Page 2/3: Kenyan Coffee Flavor Wheel Analysis
- The Flavor Wheel: A sensory tool that classifies and defines the complex range of flavors and aromas found in coffee, organized hierarchically from general categories (e.g., Fruity) to specific notes (e.g., Blackberry, Lime).
- The Signature Kenyan Profile: Kenyan coffees are typically placed in the Fruity and Sour/Fermented categories (due to high acidity) and often show secondary notes in the Floral and Sweet categories.
- Key Flavor Descriptors and Their Chemical Basis: | Flavor Descriptor | Chemical Basis | Roasting Effect | | :— | :— | :— | | Blackcurrant/Berry | High Malic Acid, specific Aldehydes | Preserved (Light Roast) | | Citrus/Lemon | High Citric Acid, high acidity level | Preserved (Light Roast) | | Winey/Tomatoes | Lactic/Acetic Acid (Fermentation by-products), Phosphoric Acid | Well-managed Fermentation/Minimal Roast | | Floral | Specific text{Strecker} Aldehydes (e.g., Phenylacetaldehyde) | Highly volatile; destroyed by dark roast | | Caramel/Toffee | Furan formation (Maillard), Sucrose caramelization | Developed (Medium Roast) |
- Using the Wheel in Cupping: The wheel serves as a standardized vocabulary. When a cupper identifies a “Blackberry” note, they are chemically identifying a blend of Malic Acid and a specific Strecker Aldehyde that survived the roast. This links the subjective experience to objective chemical science.
Page 3/3: Practical: Blind Cupping of Different Roast Profiles
- Objective: To integrate all sensory, chemistry, and technical skills by blindly evaluating the final product.
- Samples: Cupping three identical Kenyan green coffees, roasted using three distinct profiles developed throughout the course:
- Light Roast (High Acidity/Fast DTR): Maximized preservation of Citric/Malic Acid and Floral Aldehydes.
- Medium Roast (Balanced/Ideal DTR): Optimal balance of Maillard sweetness, limited caramelization, and moderate acidity.
- Dark Roast (Long DTR/Slow Cool): Maximized pyrolysis, high Quinic Acid/Pyrazines, minimal desirable acids.
- Cupping Procedure:
- Aroma (Dry/Wet): Identify text{Strecker} products (floral vs. roasty).
- Flavor/Acidity: Evaluate the quality and intensity of the acids (sharp/sour vs. bright/complex vs. flat/bitter).
- Sweetness/Body: Judge the intensity of Maillard/Caramelization products (clean/candy vs. heavy/molasses).
- Overall Balance: Determine which cup achieves the desired complex and balanced Kenyan profile.
- KCS PROJECTION : This final lab is the proof-of-concept for the entire course. The student is expected to taste the difference caused by the chemical decisions made at every stage—from soil (Day 9) to roasting (Days 11-15) to cooling (Day 16) and aging (Day 17). Mastery is achieved when the student can confidently state, “This cup is bitter and harsh because the text{ROR} was too high, leading to excessive pyrolysis and Quinic Acid formation.”